
By this means Thomson measured the ratio of the charge to the mass of the "rays,” and established that they were particles, much lighter, and presumably much smaller, than atoms. The one with the greatest import, both for his research and for experimental techniques in the emergent field of atomic physics, effected the deflection of the cathode rays by electric and magnetic fields simultaneously. Thomson devised many discharge tubes in his study of cathode rays. Thomson, Director of the Cavendish Laboratory, Cambridge University, produced convincing evidence that cathode rays were universal sub-atomic constituents of matter, of identical mass and electric charge. (See object ID EM.N-08517, Discovery of electron display apparatus with cathode ray discharge tube.)īetween 18 J. Philip Lenard, working in Germany, greatly intensified the physicists' interest in these "cathode rays" by bringing them out of the discharge tube through a hole sealed with metal foil. The discovery of electron led by sir J.J Thomson through cathode ray tube experiment is one of the most well received experiment in physics. In his experiment, he placed two plates of opposite charges near the cathode rays. Thomson began experimenting with cathode ray discharge tubes and studied the properties of the cathode ray. In the early 1890s attention focused upon the rays which, at the highest evacuations, stream out from the negative pole (cathode) of the discharge tube and produce a fluorescent glow of its glass walls. In 1897, Joseph John Thomson, a British physicist proved that atoms are not the fundamental unit of matter. Thomson ( Cambridge Eng- tion of cathode rays. (See object ID EM.N-08253, Geissler tube demonstration apparatus.) that Lenar rays are simply a secondary propagaCathode Rays.-J. The development of theory and experimental technique raised expectations of insight from this quarter into the structure of the atom and the connection between matter and electricity. In the second half of the nineteenth century the phenomena accompanying the discharge of electricity through highly rarified gases received increasing attention from physicists. Thomson - the Centenary of His Discovery of the Electron and his invention of Mass Spectrometry, Rapid Communications in Mass Spectrometry, Vol.11, 2-16 (1997). Thomson, the Cavendish Laboratory, and Thompson's cathode ray tube and positive ray apparatus, see J.J. Used to determine the ratio of charge to mass (q/m) of the electron. External magnetic field can be applied to balance electric deflection. Metal plates can have electric field applied across them. 
Rays from cathode in cylinder pass through 2 metal slots and between 2 metal plates onto glass ball. Four shorter glass tubes with internal wires extend perpendicularly from the cylinderical body (see 1st object on left in accompanying image). This object consists of a glass cylindrical body with a larger diameter glass ball at one end. Object ID EM.N-08013-A overall length 30.5 cm, glass ball diam. Thomson's cathode ray tube #2, replica of Cavendish Lab apparatus. Everett who helped to greatly increase Thomson's experimental range.J.J. About 1894 he acquired an excellent glassblower named E. He was very fumble fingered and had a tendancy to break things. Incidently, Thomson was a very unhandy person. The amount the cathode ray bent from the straight line using either the electric field or the magnetic field allowed Thomson to calculate the e/m ratio. Cathode rays move opposite to anode rays. Later it was found that anode rays and Cathode rays both exist at the same time. Rutherford model could not explain stability of atom because electron will. JJ Thomson did an experiment with discharge tube using hydrogen gas. This allowed him to use either electrical or magnetic or a combination of both to cause the cathode ray to bend. Thomson discovered electron with the help of cathode ray experiment. Thomson also could use magnets, which were placed on either side of the straight portion of the tube just to the right of the electrical plates. Thompson, conducted the cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from. 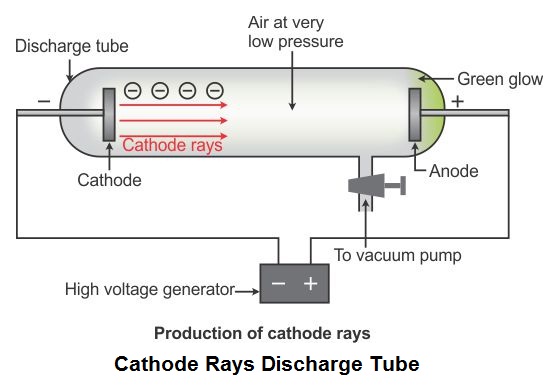
The two plates about midway in the CRT were connected to a powerful electric battery thereby creating a strong electrical field through which the cathode rays passed. The long glass finger (in the photo) projecting downward from the right-hand globe is where the entire tube was evacuated down to as good as a vacuum as could be produced, then sealed. Thomson in 1897 announcing the discovery of the electron. Th diagram below appeared in an article by J.J. It is about one meter in length and was made entirely by hand. 
The image below of a CRT used by Thomson in his experiments. Only the end of the CRT can be seen to the right-hand side of the picture. Thomson and a cathode ray tube from around 1897, the year he announced the discovery of the electron. Thomson used results from cathode ray tube (commonly abbreviated CRT) experiments to discover the electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
